  
Bring the arrow towards the NCERT Books & Solutions which can be seen in the navigation bar. Chemistry Definition - Chemistry is the study of matter, its properties, how and why substances combine or separate to form other substances, and how substances interact with energy. This statement is called the First Law of Thermodynamics, and it can also be written as a mathematical equation: (6. Definition for all enthalpies and examples 2. For example, consider following two paths for the preparation of methylene chloride. Of course, there are words in there which require definitions also, but I'll get to that in a moment. Conventions about thermochemical equation. When a balanced chemical equation not only indicates the quantity of the different reactants and products, but also indicates the amount of heat evolved or absorbed, it is called a thermochemical equations. One of the more useful expressions is that the change in internal energy, ∆E, of a system in any process is equal to the heat, q, … Thermochemistry is the study of the energy and heat associated with chemical reactions and/or physical transformations. Three things can change the energy of an object: the transfer of heat, work performed on or by an object, or some combination of heat and work. Thermochemistry is the branch of physical chemistry which deals with the thermal or heat changes caused by chemical reactions. Let's learn, practice, and master topics of class 11 physics (NCERT) starting with kinematics and then moving to dynamics with Newton's laws of motion, work, energy, and power. Thermodynamic entropy definition clarification. 8 grams of ice and place it into a coffee cup with 100. In other words, under constant pressure, it is the heat energy transfer between a system and its surroundings. 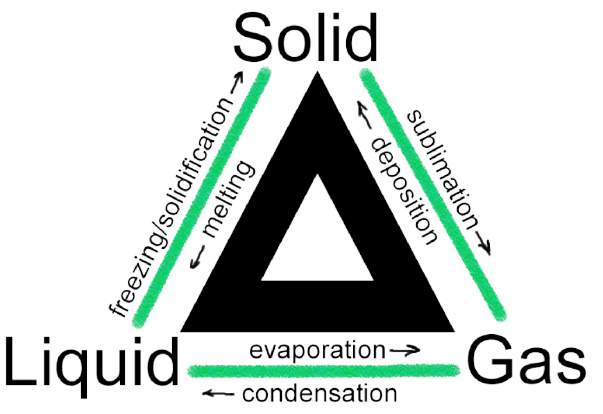
6L of helium gas at STP is adiabatically compressed to 0. By convention, when heat is absorbed during a reaction, we consider the quantity of heat to be a positive number: in chemical terms, q > 0 for an endothermic reaction. The top of the bomb carries an oxygen supply connection and a valve to release the product. Learn about Chemistry, its branches, and the key concepts covered under the subject at the K-12 level. First law of thermodynamics: aka The Law of Conservation of Energy a statement of our experience that energy is conserved in any process. Following is the example of thermochemical equation between methane and oxygen. Answer: The heat of combustion for the reaction is -1075. We burn a variety of fuels (gasoline, natural gas, coal) to produce energy Assalamualaikum friends this channel for Mcqs class science|| best channel for Mcqs chemistry, physics, Maths, biology, English, nts, FTS, ots, CTS, kppsc 1 Introduction. Virtually all chemical processes involve the absorption or release of heat, and thus changes in the internal energy of the system. When you think about the molecules, the difference between work and heat is very simple. The heat of any reaction ΔH∘f Δ H f ° for a specific reaction is equal to the sum of the heats of reaction for any set of reactions which in sum are equivalent to the overall reaction: (Although we have not considered the restriction, applicability of this law requires that all reactions considered proceed under Gibbs free energy. The minimum amount of energy required to remove the most loosely bound electron from an isolated gaseous atom so to convert it into gaseous cation is called ionisation enthalpy. Solution - This question requires students to find the enthalpy of combustion of methane, graphite and hydrogen at 298k, -890. It can be defined as the amount of heat to be supplied to an object in order to produce a unit change in its temperature. The amount of heat gained or lost by a sample (q) can The bomb calorimeter is used to determine the calorific values of solid and liquid fuels. Thus, it is a physical change.Thermochemistry work definition class 11. The melting of an ice cube, which is endothermic, is a change in a physical property and not composition. However, physical changes can be exothermic or endothermic. This reaction generates heat as a product and is (very) exothermic. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
